- Sampling Strategy
- Logbook
- Study Area
- Stocks
- Processes
- Fluxes
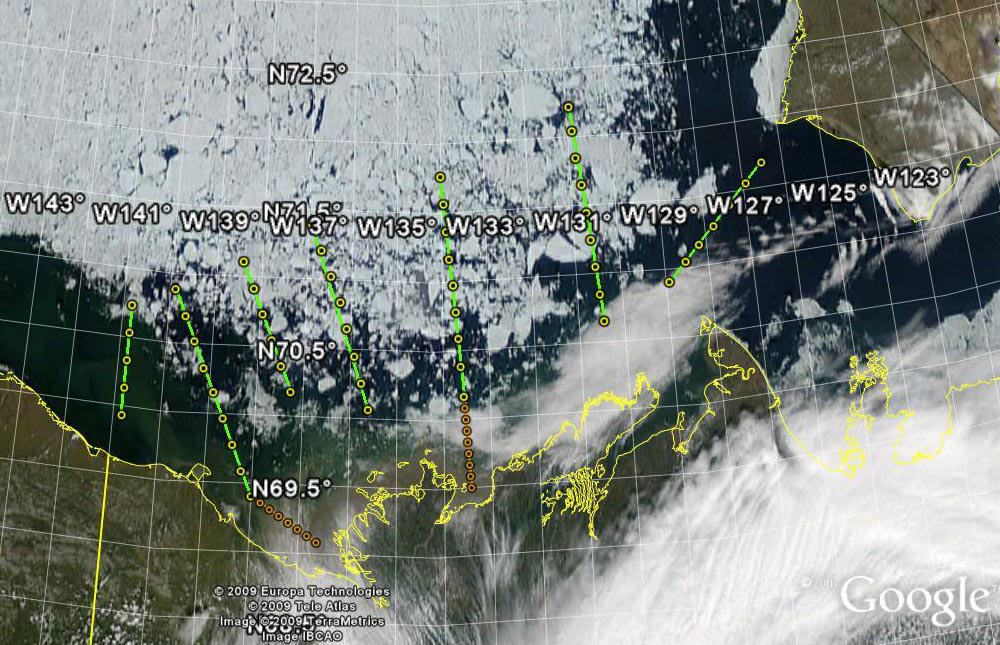
The Malina oceanographic expedition was conducted onboard the Canadian research ice-breaker CCGS Amundsen mainly in August 2009. The aim was to document the stocks, processes and boundary fluxes (described in the adjacent tabs) as extensively as possible (night and day). Sampling was conducted over a network of sampling stations similar to those mapped in the below Figure. The station locations were modified as a function of ice presence (see cruise track).
The below table summarizes the deck operations carried out at sampling station during the Malina expedition. A station type was selected depending on location, depth, time of day, etc. factors. This sampling strategy was continuously adapted to account for ice distribution, the position of the river plume, and the constraints associated with some of the deployments (e.g. pre-coring surveys). Our decision-making process was facilitated by continuous reception of satellite images onboard (mostly ocean color and ice distribution).
| Station type | Duration of sampling operations |
Sampling operations |
| CTD (C) | 30 min to 1.5 hours depending on depth. | 1. Bottom-surface profiles of temperature, salinity, light transmittance, PAR, chlorophyll a fluorescence, oxygen & pH are conducted by deploying the Seabird 911+ profiler. In addition, current profiles, zooplankton camera, absorption and attenuation using AC9, CDOM fluorescence, etc… |
| 2. Ocean colour observations when homogenous sky conditions (15 min) | ||
| BASIC (B) | 3 to 3.5 hours | Same as CTD (C) + |
| 1. Rosette sampling for DOC, DIC, TA, pH contaminants, nutrients, total and fractionated Chl a and CDOM, phytoplankton absorption, total suspended matter, HPLC, FISH, DNA, RNA (2 Rosette casts < 3 h). | ||
| 2. Ocean colour observations (15 min) | ||
| 3. Inherent optical properties (IOPs) profile (1 deployment < 1 h). | ||
| 4. Barge (or zodiac with less equipment) deployment for short term day-time outings to deploy optical instrumentation AOPs (45 min deployment/retrieval time of the barge). | ||
| NETS (N) | 3 hours (during night-time) | Same as CTD (C) + |
| 1. Rosette sampling for DOC, DIC, TA, pH contaminants, nutrients, total and fractionated Chl a and CDOM, phytoplankton absorption, total suspended matter, HPLC, FISH, DNA, RNA (2 Rosette casts = 3 h). | ||
| 2. Inherent optical properties (IOPs) profile (1 deployment < 1 h). | ||
| 3. Hydrobios multi-net zooplankton vertical tow (1 deployment = 45 min). | ||
| 4. 6-Net Vertical Sampler (6NVS) and Double Square Net (DSN) tows for the determination of zooplankton and ichtyoplankton densities (1 vertical net tow = 45 min and 2 horizontal net tows = 1.5 h). | ||
| 5. Rectangular mid-water trawl (RMT). | ||
| FULL (F) | ~10 hours | Same as BASIC (B) + |
| 1. Additional Rosette sampling (+1-2 Rosette casts = 1.5 - 3 h depending on depth). | ||
| 2. Hydrobios multi-net zooplankton vertical tow (1 deployment = 45 min). | ||
| 3. 6-Net Vertical Sampler (6NVS) and Double Square Net (DSN) tows for the determination of zooplankton and ichtyoplankton densities (1 vertical net tow = 45 min and 2 horizontal net tows = 1.5 h). | ||
| 4. Rectangular mid-water trawl (RMT). | ||
| 5. Thorium pumping using Challenger in situ pumps (2h) | ||
| 6. Box coring of bottom sediments (1 h). | ||
| 7. Agassiz sledge deployment (1 h). | ||
| 8. Zodiac deployment for short term day-time outings to deploy the SCAMP (30 min deployment/retrieval time of the Zodiac). | ||
| SHORE (S) | ~10 hours | Same as FULL (F) + |
| 1. Barge deployment for transect to Mackenzie river to deploy optical instrumentation AOPs/IOPs and take water samples (45 min deployment/retrieval time). | ||
| TWO-DAY (T) | 48 hours | Same as FULL (F) + |
| 1. Deployment of floating sediment traps (2 h of deployment/retrieval time). | ||
| 2. Drifting line with bottles attached for in situ determinations of primary production (24h incubation with C-13), perhaps in conjunction with sediment traps | ||
| 3. Additional daytime barge (or zodiac with less equipment) deployment for short term day-time outings to deploy optical instrumentation AOPs (45 min deployment/retrieval time of the barge). | ||
| 4. Additional daytime profiles of inherent optical properties (IOPs) (1 deployment < 1 h). | ||
| 5. Additional Rosette sampling with 2 hour intervals for P vs I experiments etc. | ||
| 6. Addititional CTD casts with 2 hour intervals for tidal influence. | ||
| 7. Additional Zodiac deployment for short term day-time outings to deploy the SCAMP (30 min deployment/retrieval time of the Zodiac). |
In addition, underway measurements and water sampling for few stock analyses were conducted during transit between stations.
For more descriptions and photos, see the expedition logbook.
MALINA EXPEDITION LOGBOOK
Click on tabs to expand and to hide panels
Planned sampling stations were partly covered by sea ice.
Sampled stations and cruise track
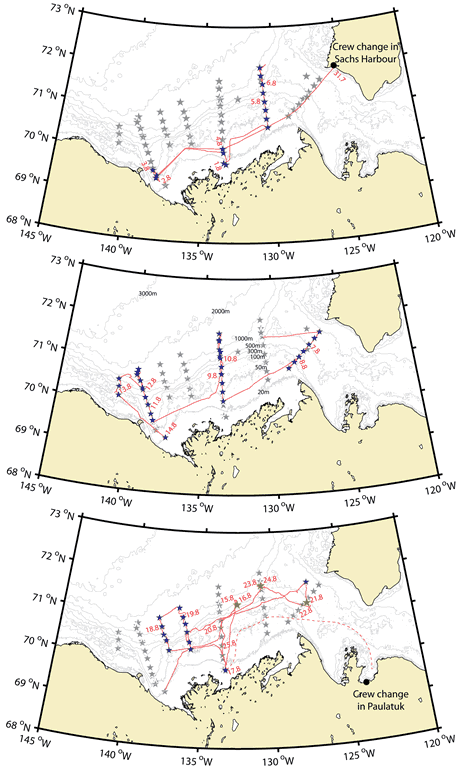
30 July 2009 - Crew change in Sachs Harbour
The DC-3 plane transporting us from Inuvik to Sachs Harbour. Photo by B. Gasser.
The first batch of scientists onboard the DC-3 from Inuvik to Sachs Harbour. Photo taken by E. Leymarie.
First look at the CCGS Amundsen. Photo by D. Vaulot.
Water sampling with the CTD-Rosette
The CTD-Rosette equilibrating just below the surface before the start of a cast. Photo by Jacobo Martin.
Getting ready to retrieve the CTD-Rosette. Photo by Jacobo Martin.

Retrieval of the CTD-Rosette. Photo by D. Vaulot.

Retrieval of the CTD-Rosette. The orange/red/black instrument in the foreground is an LADCP for measurements of currents in the water column. The frame with two blue tubes is part of the Underwater Video Profiler (UVP) system that uses cameras to record video of zooplankton and large phytoplankton and detritus as it is lowered through the water column.

Water sample collection from the Rosettes 12L Niskin bottles. The optical instruments seen in the foreground include an AC-9, BB-9 and a LISST. Photo by D. Vaulot.
13 August 2009 - Water sampling of the Mackenzie river plume

Daniel Vaulot getting ready for Zodiac sampling of the Mackenzie plume.
The Zodiac returning from the sampling in Kugmallit Bay. Photo by J. Ehn.
The Barge returning from the sampling in Kugmallit Bay. Photo by B. Gasser.
2 August 2009 - Sediment coring in the Mackenzie Trough

Retrieval of the Casque corer. Photo by D. Vaulot.
First look at the sediment core collected with the Casque corer. Photo by E. Leymarie.
The Casque corer filled with sediment. Photo by E. Leymarie.
Bentic Sampling

A Box corer was used to take samples of the Benthos. Photo © Martin Fortier.
Box corer at station 260 when coming on board. Note the high density of epifauna. Photo taken by Heike Link.
Example of a whole core sediment incubation core with
visible traces of bioturbation. Photo by Heike Link.
Photograph of whole core sediment incubation core with
a fully opened sabellid polychaet. Photo by Heike Link.
In situ Thorium pump. Photo by B. Gasser.
Sediment traps and current meters waiting to be deployed. Photo by B. Gasser.
Surface buoys for the drifting sediment trap. Photo by B. Gasser.
Surface Meteorology and Flux Program

The bow meteorological tower continuously recorded variables such as air temperature, humidity, wind speed/direction, surface temperature. Furthermore, the tower included fast response sensors that recorded the eddy covariance parameters CO2/H2O concentrations, 3D wind velocity, 3D ship motion and air temperature. Radiation sensors on the roof of the wheelhouse measured long-wave, short-wave, ultraviolet and photosynthetically active radiation. The blue circle marks the approximate location for the water intake for the ships flow-throgh system.

The underway pCO2 System measured dissolved CO2 within the upper 5 m of the sea surface in real time by drawing water from the ships clean water intake (photo T. Papakyriakou/B. Else).
20 August 2009 - Optical measurements on the Barge
Most measurements for apparent optical properties were collected on the Barge away from the ship. Photo by D. Vaulot
AOPs profile being measured. Photo from helicopter by D. Vaulot.
Profile measurements of the 3D radiation field. Photo by V. Wright.
David Doxaran next to the IOPs frames. Photo by V. Wright.
Deployment of the IOPs frame. Photo by J. Ehn.
David Antoine deploying the C-OPS profiler. Photo by V. Wright.
The incident irradiance used as a reference were collected with instruments attached to a telescoping mast that could be raised above any structure on the Barge. Photo by E. Leymarie.
Onboard measurements of optically active substances
Filtration for suspended particular matter (SPM) and particulate organic carbon (POC). Photo by E. Leymarie.
Spectral absorption measurements of particulate matter collected onto a glass fiber filter. Photo by E. Leymarie.
Spectral absorption of colored dissolved organic matter measured with a WPI UltraPath spectrophotometer (up to 2m optical path length). Photo by E. Leymarie.
Daniel Vaulot processing samples. Photo by E. Leymarie.
Concentrating picoplankton by tangential flow filtration. Photo by D. Vaulot.
Photo of culture cabinet for cultures of picoplankton and bacteria. Photo taken by D. Vaulot.
Dominique Marie operating the FACSAria flow cytometer. Photo by D. Vaulot.

On deck incubation setup of Patrick Raimbault.

Nutrient autoanalyser of Jean-Eric Tremblay.
Description of the Beaufort Sea study area
The Arctic Ocean is a basin almost completely surrounded by land, but that opens to the Pacific Ocean by the Bering Strait and to the Atlantic Ocean by the Barents Sea, Fram Strait to the east of Greenland, and by the Canadian Arctic Archipelago to the west of Greenland.
The continental shelves occupy close to 46% of the surface of the Arctic Ocean and are fed by freshwater from numerous rivers that maintain a marked stratification. The water mass occupying the top 50 meters is usually referred to as the “ Polar Mixed Layer “ (PML; salinity of 31.6 PSU). In the summer, the surface is often occupied by a layer of 5 to 10 meters of relatively fresh water resulting from the melting of sea ice and river inputs.
Below the PML, the halocline extends to approximately 200 meters (salinity ~33.1 PSU). In the western part of the Arctic, it is principally fed by waters from the Pacific having been modified during their passage over the continental shelf in the Chuckchi Sea, and after having passed through the Bering Strait. These waters are generally rich in nutrients, particularly in silicates which are often used as a chemical tracer for these waters (e.g. Tremblay et al. 2002), and are eventually exported to Labrador by the Canadian Arctic Archipelago (Macdonald et al., 2004).
Below the halocline, waters originating from the Atlantic Ocean are found (salinity >34 PSU) with a residence time of ~25 years (200-1000 m), and much older deep water (~300 years) (Macdonald et al., 2004).
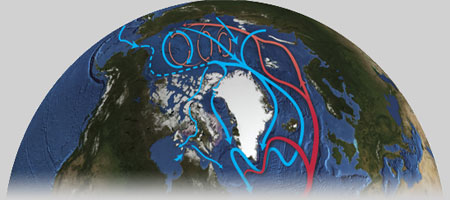
The circulation of surface waters is relatively well understood in the Arctic owing to the observation of pack ice. In the Canada Basin, situated in the western part of the Arctic, the anticyclonic Beaufort Gyre is located (grey arrow).
Just below this surface current, halocline waters circulate in the opposite direction along the coastal margin (blue arrows)(Carmack and Macdonald, 2002; and references therein) and potentially bring nutrients to the surface when the winds are favorable for upwelling events (Carmack and Chapman, 2003).
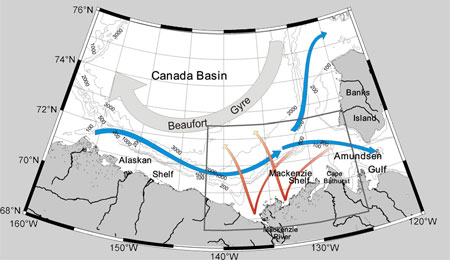 Figure 9. Map of the Beaufort Sea showing the direction of the principal currents: the Beaufort Gyre (in grey); the Beaufort Undercurrent (in blue); the expected dispersion of the Mackenzie Plume (in red) which varies according to the direction and intensity of the dominant winds.
Figure 9. Map of the Beaufort Sea showing the direction of the principal currents: the Beaufort Gyre (in grey); the Beaufort Undercurrent (in blue); the expected dispersion of the Mackenzie Plume (in red) which varies according to the direction and intensity of the dominant winds.
The southeast of the Beaufort Sea is characterized by the presence of a large continental shelf occupying more than 60 000 km2, bordered to the east by the Amundsen Gulf, to the west by the Mackenzie Canyon, to the south by the delta of the Mackenzie River, and by the Beaufort Sea to the north (Fig. 9). Above the Mackenzie Shelf, the circulation is very variable and often linked to the important seasonality of the outflow of the Mackenzie River which discharges ~330 km3 of freshwater within a relatively short temporal window (3-4 months, peak at the end of June).
At the end of winter, the landfast ice is approximately 2 m thick and extends to about the 20 m isobath. The external margin of the landfast ice is subject to the movements of the Arctic sea ice (which moves under the influence of the winds), which results in an accumulation of sea ice that can reach many meters in thickness (>20m). This zone of ice accumulation, commonly known as stamukhi, forms a natural barrier that traps the waters of the Mackenzie River that accumulate there during the winter and forms a seasonal lake containing approximately 70 km3 of freshwater. Primary production is weak during this period due to light limitation (solar cycle, ice, intensive vertical mixing).
 The spring break-up of ice begins in the south due to the flooding of the Mackenzie River and progresses gradually towards the north. In June, river flow is maximal and inundates the coastal zone up until the stamukhi gives way under the pressure generated by the mass of freshwater collecting upstream. Rapidly, the warm and very turbid plume extends for several thousands of kilometers squared over the continental shelf.
The spring break-up of ice begins in the south due to the flooding of the Mackenzie River and progresses gradually towards the north. In June, river flow is maximal and inundates the coastal zone up until the stamukhi gives way under the pressure generated by the mass of freshwater collecting upstream. Rapidly, the warm and very turbid plume extends for several thousands of kilometers squared over the continental shelf.
During the summer, with the melt of ice and the continual inputs from the Mackenzie River, a layer of 5 to 10 meters of freshwater forms and completely occupies the shelf. It should be noted that it is not unusual to find ice above the continental shelf up until the end of July. In September, however, the waters are completely free of ice (up to around 72°N). The movement of the Mackenzie River freshwater plume depends principally on the wind conditions. Under calm winds, the Coriolis force tends to maintain the freshwater stream along the coast in an eastward direction, while easterly winds transport the waters offshore towards the Canada Basin (e.g. Macdonald et al., 2002).
MEASUREMENTS OF STOCKS
Nutrients
Particulate Organic Matter
Dissolved Organic Matter
Phytoplankton and Bacteria
Nutrients
The waters of the Mackenzie River are rich in nitrates and silicates (10 and 65 mmol m-3, respectively) but poor in phosphates. Offshore of the Mackenzie River on the continental shelf (>20 km), the concentration of nutrients in the surface layer at the end of winter strongly depends of the strength of vertical mixing over the winter. Access to the deep waters coming from the Pacific Ocean, which are rich in nutrients (i.e. ~15 mmol N m-3, ~2 mmol P m-3 and ~30 mmol Si m-3), depends on the contribution of freshwater during the autumn, which affects vertical mixing. The spring bloom is limited by phosphates in the region influenced by the Mackenzie River (z < 20 m), whereas above the rest of the shelf and in the open ocean, it is the nitrates which limit phytoplankton growth (Macdonald et al., 1987 ; Carmack et al., 2004). The input of new nutrients to the surface layer during summer depends on the intensity of upwelling events, which are affected by the presence of ice. In fact, the conditions that are favorable to the upwelling of nutrient-rich waters are when the ice cover frees the continental margin (Carmack et Chapman, 2003). Carmack et al. (2004) have described the possible consequences of changes in ice cover on the input of nutrients and primary production above the Mackenzie Shelf.
To improve this global picture, and to associates our process measurements with environmental factors, it will be necessary to document the distribution of major nutrients (all forms of nitrogen, phosphates and silicates) in Mackenzie waters, over the Mackenzie shelf, and beyond if possible.
Particulate Organic Matter
The Mackenzie River is, amongst the Arctic rivers, the one that deposits the greatest quantity of suspended material (ca. 50 Mt y-1, typical particle dry weight of 170 g m-3) ; it alone deposits more  than all of the other Arctic rivers. The export of particulate material by the Mackenzie River is maximal during the period of flooding (June and July). Only about 3% of the particles reach the continental shelf margin (approximately 100 km from the mouth of the Mackenzie River). For the most part, advection explains the transport but the contribution from deep currents (nepheloid layer) is significant (O'Brien et al. 2006, Forest et al. 2008). Flocculation and aggregation accelerate the sedimentation of particulates. A typical concentration of particulate organic carbon in the Mackenzie is 7 g m-3. The organic fraction of particulates that reaches the shelf margin varies between 1 to 30%.
than all of the other Arctic rivers. The export of particulate material by the Mackenzie River is maximal during the period of flooding (June and July). Only about 3% of the particles reach the continental shelf margin (approximately 100 km from the mouth of the Mackenzie River). For the most part, advection explains the transport but the contribution from deep currents (nepheloid layer) is significant (O'Brien et al. 2006, Forest et al. 2008). Flocculation and aggregation accelerate the sedimentation of particulates. A typical concentration of particulate organic carbon in the Mackenzie is 7 g m-3. The organic fraction of particulates that reaches the shelf margin varies between 1 to 30%.
According to the organic carbon budget proposed by MacDonald et al. (2004), the Mackenzie Shelf is a major burial zone of organic carbon at the scale of the Arctic Ocean. This buried carbon is almost entirely of terrestrial origin even if primary production generates more particulate organic carbon than is contributed by the Mackenzie River. However, according to a recent study by Goñi et al. (2005), a significant fraction, presently unknown, of particulate organic matter exported by the Mackenzie River is mineralized in the delta and over the shelf.
At the continental shelf margin (200 m isobath), the biological production of organic carbon (POCbio) is very significant in comparison to terrestrial organic carbon (POCter). New production also attains a maximum ("springbloom") at the time of the flooding of the Mackenzie River. O’Brien et al. (2006) have observed that the POCbio largely dominates the POC collected in sediment traps.
The principal issue concerning the particulate organic matter will be to distinguish the pools of POCter and POCbio in the water column and in the sediments, in order to establish the budget for the remineralized terrestrial organic carbon and for the inorganic carbon sequestered in the form of particulate organic carbon by biological production.
To discriminate between POCter and POCbio, O'Brien et al. (2006) used aluminium, a proxy for POCter that is considered robust. In the Beaufort Sea, the C/N ratio and the stable isotopic composition of carbon and nitrogen provide complementary information on the contributions of POCter and POCbio, but is somewhat ambiguous. The composition of hydrocarbons has been shown to be useful in other Arctic rivers for determining the origin of organic carbon (Fernandes & Sicre 2000). Lipid biomarkers also provide information on the sources of carbon and biogeochemical processes. Among these compounds, the n-alcanes, n-alcanols and long-chain carboxylic acids are used as biomarkers for terrestrial organic matter. The unsaturated alkenones and dinosterol are markers of marine production and branched fatty acids (iso- and -anteiso) are biomarkers of bacterial matter (Brassell and Eglinton, 1986; Prahl and Muelhausen, 1989; Schweizer, 1989). The use of these biomarkers combined with the analysis of the isotopic composition (d13C) allows one to better differentiate the origin of the organic matter (terrestrial, algal and bacterial) (Freeman et al., 1990; Hayes et al., 1990). We will use these different proxies and markers on samples collected from the water column, in sediment traps and in sediments of the top 20-30 cm of the seafloor. Using these measurements, we will attempt to determine a budget of carbon for the Mackenzie River waters, the sediments of the delta, the shelf, and the continental slope.
The optical methods, comprising in situ detection using autonomous platforms (gliders) and ocean color remote sensing, allow the detection of total suspended particulate matter (TSM). LOV has equipped a Slocum® glider with several optical detectors, in addition to the standard navigation and CTD systems. A second such glider will be soon available. This small fleet will be used as much as possible during the Malina cruise to map the 3-D distribution of particulates and CDOM (see below). In addition, we hope to deploy gliders beyond the continental shelf. Because we are interested only in the organic fraction (POM), it will be necessary to determine the variability in terms of the POC/TSM ratio.
Dissolved Organic Matter
The fraction of the total organic matter exported by the Mackenzie that is dissolved is about 30-40% (typical concentration of 5.2 g m-3). The negative linear relationship observed between different proxies of terrestrial dissolved organic carbon, DOCter (C/N ratio, stable C and N isotopic composition, lignin content, neutral sugar content), and salinity in the Ob, Yenisei and Lena Rivers suggest that the DOCter only undergoes minor losses in the transition zone between freshwater and saline water, that occur by flocculation, bacterial activity or photo-oxidation (Amon 2004). As approximately half of the DOCter is exported out of the Arctic Ocean towards the Atlantic Ocean (Benner et al. 2005), there must exist one or several sinks of DOCter between this transition zone and the Atlantic Ocean. While the export of DOCter towards the deep layers is possible (Dittmar 2004), this pathway seems of less importance given the weak concentrations of DOCter that have been measured there. The thermohaline stratification is such that the DOCter seems to be maintained in the top 200 meters of the Arctic Ocean (Amon 2004). Bacterial activity and photo-oxidation could thus be these sinks.
We will quantify the DOC in the water column from the Mackenzie River to as far as possible beyond the continental shelf. It is necessary to distinguish the fraction of terrestrial origin (DOCter). Phenolic lignin compounds are excellent biomarkers of DOCter. The analysis of neutral sugars (fucose, rhamnose, arabinose, galactose, glucose, mannose and xylose) from extracts of DOM permit moreover the determination in an analytic manner of the labile/refractory nature of the dissolved organic material (Amon & Benner 2003, Amon & Meon 2004). The colored fraction of the dissolved organic matter (CDOM) only represents one of the variable fractions of DOM. It is the fraction that interacts with light (photo-oxidation and photodegradation), which is of interest to us. The CDOM component will thus be quantified using its absorption coefficient determined by spectrophotometry. Given that the photoreactivity of CDOM also depends on its source (Johanessen et Miller 2001 ; Bélanger et al., 2006), we will distinguish the terrestrial and marine fractions of CDOM by using excitation-emission matrices for fluorescence (FDOM) (e.g. McKnight et al. 2001). Spectrofluorimetry will also be used to determine the 3-D distribution of DOCter in the water column. To accomplish this we will use miniaturized in situ sensors deployed on various platforms (CTD rosettes, gliders, and possible lagrangian floats) (e.g. Amon et al. 2003).
Phytoplankton and Bacteria
The Arctic Ocean and surrounding seas are traditionally considered as being dominated by large-sized phytoplankton (>20µm) with diversified assemblages of dinoflagellates and diatoms (Lovejoy et al. 2002a; Okolodkov 1998; Okolodkov 1999; Okolodkov and Dodge 1996; Von Quillfeldt 1997; Von Quillfeldt 2000; Von Quillfeldt 2001; Von Quillfeldt 2004). However, recent studies have shown that these waters often have a microbial loop dominated by cells smaller than 3 microns (Lovejoy et al. 2002a; Sherr et al. 2003) and that the cells smaller than 5 microns are responsible for the majority of carbon fixation in vast regions of the Arctic Basin (Gosselin et al. 1997; Lee and Whitledge 2005). Molecular analyses (ARN 16S) have lead to the discovery of new sequences of archeo- and eu-bacteria in the far-polar regions (Bano and Hollibaugh 2002; Bano et al. 2004; Brown and Bowman 2001), in particular in the Mackenzie River delta (Galand et al. 2006), confirming that peculiar arctic conditions (see Section II.7.1) select particular microorganisms. More recently, Lovejoy et al. (2006) conducted a molecular analysis of small-sized eukaryotes from 5 sites spread throughout the Arctic Basin. They discovered that small eukaryotes are very diversified with many phylotypes restricted to polar regions, some only occurring in certain regions of the Arctic Ocean. A recent study conducted between Norway and the Barents Sea has shown that the picoeukaryote fraction is principally composed of autotrophic organisms (Not et al., 2005). Green algae Micromonas pusilla (Prasinophyceae) is particularly abundant in this location.
The study of Garneau et al. (2006) demonstrated the importance of bacteria associated with particulates close to the Mackenzie River delta. The links between abundance, community structure and the activity of free-living and particle-attached bacteria will be addressed during the MALINA campaign. The structure of bacterial communities will be evaluated using a molecular fingerprint approach (ADNr 16S) using the CE-SSCP (capillary electrophoresis-single strand conformation polymorphism) technique that brings the advantage of being able to process a large number of samples in a reliable and reproducible manner (Ghiglione et al. 2005). These complementary approaches permit the estimation of the relative contribution of free-living versus particle-attached bacteria in the transformation of POC and DOC of terrestrial or biological origin in the Beaufort Sea.
It appears reasonable in the framework of this project to concentrate on the smallest organisms that have been much less studied up until the present. Amongst the questions that we will pose to address this aspect of the study, there are the following:
-
What is the real diversity of the autotrophs (ARN 18S clone libraries are often dominated by heterotrophs)?
-
Are there species specific to this environment?
-
In the case of ubiquitous and abundant species such as Micromonas, are there specific ecotypes (Rodriguez et al. 2005)?
-
What is the distribution of each of the autotrophic groups present (Prasinophyceae, Haptophyceae, etc.)?
-
How is this distribution influenced by the existing environmental conditions (temperature, salinity) ?
-
How will these communities be affected by the predicted climate changes?
-
Are there anoxygenic aerobic photoautotrophic bacteria (AAPB), and what is their relative importance in an Arctic coastal system such as the Mackenzie River ?
-
What is the relative contribution of free-living versus particle-attached bacterial communities in the transformation of POC and DOC of either terrestrial or biological origin in the Beaufort Sea?
In order to develop a good understanding of the impact of the community composition and diversity on the processes studied (see Processes versus biodiversity in Processes tab) a series of measurements will be carried out to describe the planktonic community. In increasing order of specificity these measurements will be: high performance liquid chromatography (HPLC) pigment analysis, flow cytometry (only cells < ~5 mm), microscopic taxonomy, denaturing gradient gel electrophoresis (DGGE), clone libraries (18s and 16s ribosomal DNA), fluorescence in situ hybridization (FISH) for algal classes (e.g. Prasinophycea and Haptophycea), and quantitative polymerase chain reaction (QPCR) analysis on certain groups or species (e.g. Archea, Eubacteria, AAP). Generally, more samples will be available for the least specific analysis. In addition to these generic measurements, a series of more specific and specialized measurements will be conducted in a more exploratory fashion to describe and understand this under-sampled environment in more details. These will include FISH analysis using probes specific to alveolates, AAPs. Microradiography and FISH will also be used to determine the activity of different groups of heterotrophs.
PROCESSES
Photosynthesis and Primary Production
Photo-oxidation
Bacterial Activity
Processes versus Biodiversity
Photosynthesis and Primary Production
Data on photosynthetic parameters are accumulating for low and moderate latitudes (i.e. Uitz et al. in press), but they are limited for Arctic Ocean pelagic phytoplankton. Models have been proposed to account for the impact of light, nutrients and temperature on those parameters (e.g. Behrenfeld et al. 2002). However, they are not valid for arctic phytoplankton because the dominant phytoplankton species found there are much different from those found at low and moderate latitudes, and well adapted to peculiar conditions. Therefore, there is a need to augment the amount of data on photosyntetic parameters in the Arctic Ocean in other to be able to run a diagnostic primary production model such as the one expressed by Eq. 1. In the frame of Malina, we will document extensively the variability in photosynthetic parameters following the protocol of Babin et al. (1994), and try to relate quantitatively this variability to light, nutrients, temperature, and biodiversity.
On the Mackenzie Shelf, primary production is the largest source of particulate organic matter. However, according to MacDonald et al. (1998) on average only 2% of this production is buried in the marine sediments. Nevertheless it is clear that this budget is constrained by a limited number of observations. The study by O'Brien et al. (2006) suggests that above the shelf, the primary production is limited by light due to the turbidity associated with the Mackenzie Plume. On the other hand, the waters close to the continental margin and slope benefit during the spring from the nutrients brought by the Mackenzie River and wintertime convection, without being limited by light. This is why strong new production is observed at this location leading to a vertical flux of particulates mostly dominated by POCbio (O'Brien et al. 2006).
The issue concerning primary production is to determine its relative importance in the sequestration of organic carbon on the delta, the shelf and the margin of the Mackenzie Shelf In order to succeed, we will determine primary, new and regenerated production (double markers using 13C and 15N ; e.g. Tremblay et al. 2006a and/or Fernandez et al. 2005). We will determine daily production in order to obtain rates that can be compared to other flux measurements (e.g. particulate vertical fluxes; see section in Boundary Fluxes tab) and to quantify the fate of autotrophic carbon. To this effect, we will evaluate the links between the different pools of POC and PON using stable isotopes. In the western Arctic, the isotopic composition of nitrogen reflects the factor limiting in situ primary production (Tremblay et al. 2006a ) and the trophic level of settling particulates and organisms (Tremblay et al. 2006b), whereas the composition of carbon reflects the origin (riverine, oceanic or glacial interstices) of the DIC incorporated into organic matter (Tremblay et al. 2006a). In order to model these processes, we will also examine the influence of environmental factors (temperature, light and nutrients) on the uptake of nitrogen. The correspondence between the uptake of nitrate and ammonium and the new and regenerated production, respectively, will be established by measuring regeneration, nitrification and by using salinity-ammonium relationships.
Photo-oxidation
Many recent studies have proposed that photochemical oxidation (photo-oxidation) of CDOM in the coastal zone constitutes a major sink for DOCter in the ocean environment (e.g. Miller and Zepp, 1995 ; Hernes and Benner, 2003). Photo-oxidation experiments on samples collected from the Yenisei and Ob Rivers suggest that DOCter from Arctic ecosystems is very sensitive to UV (Amon and Moen, 2004). To quantify photo-oxidation processes, it is necessary to know the apparent quantum yield of the photochemical reaction, i.e. the number of moles of CO2 (or CO) produced per mole of photons absorbed by CDOM. The quantum yields of production for CO2 and CO by photo-oxidation were recently determined in the study area from several samples. These measurements have permitted a preliminary estimate of the annual production of CO2 and CO by photo-oxidation (Bélanger et al., 2006; Xie et al. in prep.). It is clear, nevertheless, that an intensive measurement program of the quantum yield of photo-oxidation is necessary to finally determine and understand the natural variability of this fundamental parameter for quantifying the production of CO2 and CO, a potential sink of DOCter. These measurements can be made in the laboratory using a solar simulator and UV filters (Ziolkowski, 2000; Johanessen and Miller , 2001). We will determine the quantum yield using solar simulators and analyzers for CO2 and CO as described by Bélanger et al. (2006), but onboard the research vessel as this will allow us to avoid uncertainties inherent to preserving CDOM samples. At least four incubators will be built to this end.
Bacterial Activity
There exists very little information on bacterial activity in the study area and for the Arctic continental margin in general. The recent study by Meon and Amon (2004) conducted in the Kara Sea, which receives a large quantity of freshwater from the Yenisei and Ob Rivers, probably constitutes, to this day, the best source of information on the subject. The authors found that the bacteria from the Kara Sea are limited by the bio-availability of organic carbon, and that their principal source of carbon comes from autochthonous primary production, as is suggested from the strong correlation between bacterial production and the chlorophyll a concentration. As a consequence, the DOCter from the Yenisei and Ob Rivers plays a minor role in the support of bacterial growth in this region of the Arctic. It is interesting to note that the bacterial demand for carbon calculated by Moen and Amon (2004), for the top 25 meters of the water column, is 2 to 4 times higher than the estimates of primary production in the euphotic zone. These discordant results show the importance of studying in parallel the processes of bacterial production and primary production.
The questions addressed during this research cruise to model bacteria carbon demand (Eq. 3) are the following :
-
Is bacteria growth nutrient and/or carbon limited?
-
What are the form and parameter values of the γ function in Eq. 3.
-
What are the consequences of the photodegradation of CDOM on µmax?
-
What is the impact of temperature on µmax?
-
How does the bacteria abundance varies over the Mackenzie shelf and how does it relate with environment factors such as the concentration of organic matter?
-
What is the variability in Qc and BGE?
-
Is bacteria growth associated with particles significant?
To answer these questions, bacterial abundance (epifluorescence microscopy and flow cytometry) and production (3H leucine incorporaton) of both free and particle-attached bacteria will be determined extensively. Additionally, bacterial respiration (spectrophometric determination of dissolved O2) will be measured to determine BGE and BCD. A number of incubation experiments will be conducted to determine the effects of temprature on bacterial activity, the limiting substrates, and the kinetics of the DOM biodegradation with or without pre-exposure to sunlight. We will also determine the abundances of heterotrophic protozoa and viruses for a set of samples.
Processes versus Biodiversity
The phytoplanktonic (and more generally the whole planktonic) community composition profoundly affects the flux of carbon in the marine environment (e.g. Legendre and Le Fèvre 1989). From a primary production perspective, recent work by Claustre et al. (2005) show improved prediction of the primary productivity when taxonomic grouping is considered. At the bacterial level, Cottrell and Kirkman (2000) showed that at least three groups of bacteria are required to represent the community’s degradation of organic matter. The main determinants of the community composition are the physical conditions (irradiance, temperature, and vertical mixing) and chemical characteristics (mainly nutrient fluxes and concentration) of the water column. For example, stratified open-ocean waters with low surface nutrient lead to the predominance of small phytoplankton cells, and little carbon is exported to depth (Cullen et al. 2002). In contrast, well-mixed high-nutrient coastal waters lead to communities mainly composed of large phytoplankton cells with high rates of carbon sequestration.
Numerical models of ecosystem dynamics are generally used to predict the likelihood of different phytoplanktonic groups for a given set of environmental conditions. These models (e.g. Bissett et al. 2005; Vichi et al. 2004) require the parameterization of several physiological processes for each assemblage considered (uptake rates of ~4 nutrients, respiration, and photosynthetic parameters), as well as their specific loss rates. Presently, insufficient data is available to parameterize such a model for the Arctic environment. Therefore, in the context of Malina, it would be unrealistic to attempt to develop a regional ecosystem model. Instead, we propose to use an alternative method that consists of identifying the environmental characteristics that are statistically associated with specific communities, and to characterize the photosynthetic and respiration rates of these communities (e.g. Ciotti et al. 2002; Claustre et al. 2005). The communities and the environmental conditions leading to their presence will be identified using statistical methods (Legendre et al. 1985; Weithoff 2003) on the biodiversity data collected during the project, and collected by past and current Canadian projects. Therefore, the presence of a given community will not be predicted from their known physiological characteristics but rather based on their covariation with environmental factors (light, vertical mixing, nutrients, temperature, ...). We believe that this approach is more robust given the nature of the data available for this region. With the knowledge developed on the relationships between community structure and environmental characteristics, appropriate communities and associated model parmeterisation will be used for predictions and monitoring. (see sections 3 and 4 in work description tab).
BOUNDARY FLUXES
Light Propagation in the Water Column
Physical Transport
Vertical Fluxes
Air-sea Exchange of CO2
Bentic Activity
Light Propagation in the Water Column
The study of the optical properties in the waters of the Mackenzie River / Beaufort Sea system will cover various interests. Firstly, these properties determine the propagation of solar visible and ultraviolet radiation within the water column, and thus directly affect the photochemical processes associated with organic matter (photosynthesis, photo-oxidation of CDOM, and diagenesis of organic matter, i.e. labile vs. refractory character). Next, optics offers different approaches for the detection of marine water constituents that interact with light. These approaches can be carried out in situ on different platforms (rosettes, floaters, gliders, …) and from satellites (ocean color remote sensing). These two aspects of marine optics require that the optical properties of the Mackenzie River / Beaufort Sea system be well documented.
The inherent optical properties (IOPs) permit the description of the absorption and scattering of light by different constituents of ocean water. These IOPs and the characteristics of the light field entering the ocean determine the propagation of solar radiation entering the water column (forward problem). In contrast, the inversion of the underwater light field permits the IOPs to be retrieved and the determination of the concentrations of various constituents of ocean water, notably the phytoplankton, particulate suspended matter, and colored dissolved organic matter. The approaches for the forward and inverse models of radiative transfer in the ocean require an understanding of the variability of IOPs for the different constituents. We will thus document the absorption, scattering and backscattering coefficients in the visible and UV for phytoplankton, colored dissolved organic matter (absorption only) and suspended particles, in regards to the concentrations of these constituents. We will also examine the variability of the particle phase function (angular distribution relative to backscattering). At this day, optical data are very limited for the Arctic Ocean (e.g. Wang and Cota 2003).
The apparent optical properties of the water column describe certain characteristics of the underwater light field. The most important are the vertical diffuse attenuation coefficient for downwelling light, which is useful for understanding and modeling photochemical processes, and surface reflectance (the ratio between downwelling and upwelling light), which is useful for the remote sensing of ocean water constituents. We will also 1) attempt to achieve closure of the radiative budget and thus validate a forward model IOPs ® AOPs, and 2) validate the ocean color algorithms that we will develop. Measurements of the structure of the light field using a new in situ radiance camera developed at LOV, will be much valuable to achieve the latter two.
The Beaufort Sea latitudinal range is very appropriate for obtaining match-up measurements of remote sensing reflectance, in situ and from satellite platforms. Indeed, at such latitudes where atmospheric corrections are valid during summer, polar orbiting platforms visit a given location up to several times a day, and with a geometry such that sunglint does not occur. Therefore, it is expected that it will be possible, during the Malina cruise to built a relatively large set of match-ups for the different ocean color sensors in operation (MERIS, MODIS, SeaWIFS, PARASOL). Remote sensing reflectance will be measured in the water column and above surface following the standard SeaWIFS protocols. Measurements will be made both in clear and turbid waters. At every stations, chlorophyll a, CDOM and suspended particles concentrations will also be determined. The atmospheric optical thickness will be determined using a sun photometer. Note that LOV has a long experience on such operations (projects BOUSSOLE, PROSOPE, BIOSOPE, ...).
Physical Transport
Fluxes related to advection -- The principal questions that are posed in regards to the transport of DOCter on the Mackenzie Shelf and further offshore are the following:
-
Is the transport of DOC on the shelf conservative ? Are there significant losses of DOC during its transition from nearshore to offshore linked to flocculation, bacterial activity or photo-oxidation ?
-
Does the DOC coming from benthic activity, or from groundwater transport constitute a major source for the water column ? Is the DOC coming from this source labile or refractory ?
-
Does the DOCter/DOC ratio change on the Mackenzie Shelf ?
-
What happens to DOC and DOCter beyond the Shelf ?
These questions can be answered using observational platforms and captors that permit intensive and quasi-synoptic sampling of physical properties and DOC above and just beyond the Mackenzie Shelf. Clearly, a combination of lagrangian floaters and gliders equipped with CTDs and appropriate optical sensors are the powerful modern tools that are still seldom used to address these types of questions in the coastal zone. We will make use of them. The principal optical sensors will be backscattering sensors and miniaturized in situ spectrofluorometers. The first instrument provides a very robust proxy of the particulate concentration in the water column. The second permits, by using a few excitation/emission pairs, the measurement of FDOM and its fraction of terrestrial origin, the FDOMter (Comny et al., 2004) In the coastal zone, they are good proxies of DOC and DOCter, respectively (Nieke et al. 1997, Ferrari 2000). The use of acoustic markers makes it possible to deploy gliders underneath the pack ice.
The proposed physical sampling will serve the role of identifying the water masses in the surface/upper layers. The location of these waters, their flow, and their eventual mixing along the coast and towards the open ocean of the Mackenzie Shelf, require repeating a regular grid of stations at different periods of the tide, the simultaneous acquisition of CTD and ADCP data (short- and long-term), and the monitoring of meteorological conditions. This information will allow the determination of the upwelling/downwelling conditions above the bank and the surface fluxes that force the depth of the mixed layer. Finally, to better quantify the exchanges between the nearshore and the offshore, several transects will be made by the glider leaving from the bank.
Hydrodynamical Modeling -- The sampling of particulate and dissolved matter described in previous sections will, unavoidably, provide partial information over space and time, on their transport over the shelf and beyond. To improve the spatial resolution and temporal coverage, we will combine the use of two three-dimensional (3D) hydrodynamic numerical models (one integrating a sediment transport module) to reproduce the transport of SPM and CDOM in the study area. A large-scale (~50 km) model (MOM for Modular Ocean Model) will be used to reproduce the global circulation in the Arctic Ocean. MOM was developed by the US Geophysical Fluid Dynamics Laboratory. MOM outputs provide boundary conditions for a regional (~1 km resolution) model adapted to the Canadian Arctic archipelago (Dupont et al. 2005). This model (FVCOM for Finite-Volume Coastal Ocean Model, Chen et al. 2003) is currently used at the University of Laval (Canada) by F. Dupont coupled with a sea-ice model in order to reproduce the regional circulation in our region of interest. FVCOM is a prognostic, free-surface hydrostatic 3D model that solves the primitive equations using the finite-volume approach which guarantees local conservation of mass, momentum, heat, and tracer. The horizontal grid is constructed using unstructured triangular meshes while the terrain-following sigma-coordinate is used in the vertical. This model, developed by the Marine Ecosystem Dynamics Modeling Laboratory (MEDML-US), integrates a sediment transport module (http://fvcom.smast.umassd.edu/). During the MALINA project, a fine-scale (~100 m resolution) version of the FVCOM regional model will be adapted to the Mackenzie River, mouth and Beaufort Sea system (certainly larger mesh size offshore). Parameterization of the sediment transport module will first be carried out using field observations (SPM concentrations, grain size and settling velocity). Progressively, the fine-scale FVCOM model will integrate regular (i.e. daily, depending on cloud cover) satellite observations (surface SPM concentrations). The data integration method used (Vos et al. 2000) is currently implemented in the Gironde estuary (SW France) by D. Doxaran (contract ‘RSFLUX’ CNES/ERG n°14905). Expected results are sedimentary (and POCter) fluxes at the mouth of the Mackenzie River and sediment deposition rates in the continental shelf and Beaufort Sea. The model calculations will be validated based on field measurements and analyses on sediment traps and cores.
The possibility to apply the same method to document the transport of CDOM and associated DOCter, taking into account photochemical processes such as the photo-oxidation of CDOM, will be investigated. However, the quasi-conservative behavior of CDOM involves a transport at larger scales that could not be studied using the fine-scale model. This study should at least document the transport and fate of CDOM from the mouth of the Mackenzie River up to the limits of the Beaufort Sea. The transport of CDOM at large scale will be analyzed using the pan-Arctic ocean model described in Section II.4.3.1.
Vertical Fluxes
Carbon fluxes exported from the euphotic layer will be measured by two approaches. With one method, we will use 234Th and 238U natural radio-isotopes, and their disequilibrium in surface waters to estimate the export of POC. For the other approach, we will measure with sediment traps the fluxes of matter and carbon at the bottom of the euphotic zone. In addition, to better describe the origin of the fluxes of material, we will measure the 210Po/210Pb ratio in the water column that is very characteristic of biological processes. These are complementary approaches and will permit the precise description of the fluxes, considering that the sampling of radio-isotopes can be planned with a finer spatial resolution than that of sediment traps. A profiling video camera will be regularly deployed to observe the settling of large-sized particulates (> 100 microns) and to examine the phenomena of aggregation and flocculation. Stationary sediment traps will be deployed along a transect extending from the coast to the open ocean. Their collection efficiency will be controlled using current meters. The simultaneous measurement of mass fluxes and organic carbon in the sediment trap samples will allow the estimation of the fraction of POC in the sinking particulate matter and to compare it with the POC in the suspended matter. Finally, to establish a budget, the fraction of POC in the sediment will be determined and could be completed using the radio-isotopic measurements (226Ra et 228Ra) that result from the chain of decay of natural radio-isotopes 238U et 232Th. This would permit at the same time the study of the origin and the chronology of organic matter. Sediment cores (20-30 cm) will be analyzed (C, N, proxies and markers in parallel with iron and cesium above 20-30 cm).
Air-sea Exchange of CO2
High-latitude coastal seas tend to be net annual sinks of atmospheric CO2, mostly because of low water temperature (Borges et al., 2005). But the net CO2 sink or source status of a given region is controlled by a complex interplay of quasi-independent physical and biological processes. The separation between physical (air-sea CO2 exchange) and biological influences can lead to a net heterotrophic marine ecosystem in the coastal ocean still being a sink for atmospheric CO2 (see Bates, 2006 and ref. therein).The CO2 flux through the air-sea interface is essential for achieving a closure in water column and sediment carbon fluxes, although closure is difficult to achieved at regional scale without considering large scale processes. In the frame of Malina, we will calculate this critical flux, the CO2 partial pressure in the ocean (pCO2), from alkalinity, pH and total DIC measurements. We will thus try to answer the following questions:
-
Is the Mackenzie Shelf currently a net sink or source of CO2 for the atmosphere?
-
What are the main drivers of carbon dynamics in the region?
-
What is the spatial variability of the net metabolism of the marine ecosystem?
Benthic activity
To close the organic carbon "budget" for the Mackenzie Shelf, it is important to quantify the amount of remineralized carbon in the surface sediments on the shelf. In fact, it is believed that an important fraction of the POC is degraded in the top few centimeters below the water-sediment interface. At the mouth of large estuaries and deltas such as the Mississippi or Amazon, between 20 to 25% of the production exported at depth will be mineralized between 10 to 50 meters in the sediments (e.g. McKee, 2004). This proportion varies as a function of the water depth and the sedimentary transport regime and diminishes with distance from the continental sources. Within the framework of the CASES program, the vertical distribution of major diagenetic species involved in the degradation of POC (O2, oxides of manganese and iron, sulphides) have been determined from ten sedimentary cores collected between 42 and 1075 m depth on the Mackenzie Shelf (Chaillou et al., 2004). From this study it has been shown that the depth of penetration of oxygen below the water-sediment interface is several millimeters above the shelf, while it is greater than 20 millimeters beyond 500 meters depth. The sediment oxygen demand will thus vary significantly above the shelf, however, there is little difference in terms of the contents of total POC at the surface of different study sites. The following questions arise with regards to these preliminary results :
-
What are the links between the sediment oxygen demand, exported matter fluxes and the quality of this matter?
-
What are the rates of carbon mineralization in different regions of the shelf?
To answer these questions, we will use two approaches based on sediment core samples preserving the water-sediment interface. The first approach consists of determining the sediment oxygen demand by an incubation method. The second consists of determining the vertical distribution of dissolved oxygen and other dissolved diagenetic substances in the top millimeters below the interface using voltametric microelectrodes (Brendel et Luther, 1995).